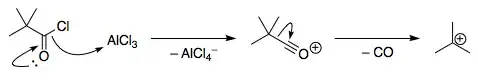
In class we were told of an anomalous Friedel–Crafts reaction with a tertiary acid chloride — $\ce{(CH3)3CCOCl}$, or pivaloyl chloride. When this reacts with benzene in the presence of $\ce{AlCl3}$, instead of effecting the normal acylation, it instead breaks down into the tert-butyl carbocation, which then alkylates benzene to form tert-butylbenzene.
I could find no satisfactory explanation for this, however, except for the fact that a tertiary carbocation is a very stable carbocation — and nor did I find any mention of this reaction on searching. Could anyone please detail this further?