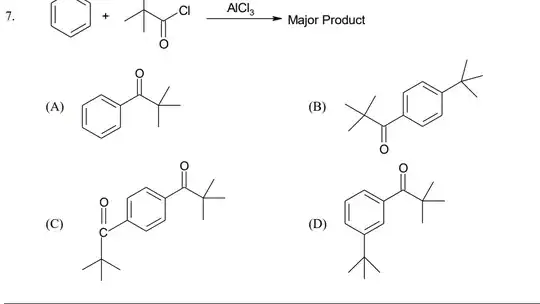
(This question is from a dubious source)
The reaction taking place here is Friedel-Crafts acylation of benzene with 1-chloro 2,2-dimethyl propanone (pivaloyl chloride?). To me, the options seem most peculiar. When the $\ce{C} - \ce{Cl}$ is broken, it gives rise to a carbocation at the carbon of $\ce{C}=\ce{O}$ bond.
According to my teacher, $\ce{C+}=\ce{O}$ now leaves with the electrons of the adjacent $\ce{C}-\ce{C}$ bond, giving rise to a stable tertiary butyl carbocation. This leads us to $\ce{PhCMe3}$.
Edit 2: this is the same reaction: Unusual Friedel–Crafts alkylation with pivaloyl chloride
Considering that the t-butyl group is para-directing, the options still don't seem right to me. If this conversion to a t-butyl carbocation happens once, can it not happen again? Would we simply not get two t-butyl groups para to each other?
(I still doubt multiple substitution is even possible)
Edit 3: as of 24/2/2024 this question has been confirmed to be of poor quality. See reasons in comments.