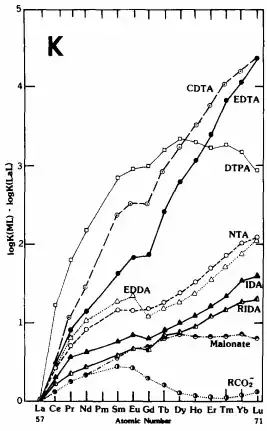
Smith and Martell obtained a series of data for the binding of trivalent lanthanide ions, $\ce{Ln^3+}$, with various carboxylic acid ligands (amongst them the well-known EDTA).1 A graph of the formation constants is attached ($K_\mathrm{f} = [\ce{Ln(edta)-}]/[\ce{Ln^3+}][\ce{edta^4-}]$):
Due to the contracted nature of the 4f orbitals, the lanthanide ions characteristically do not exhibit covalency in metal-ligand bonding, which is strongly reflected in their chemistry. Therefore, the almost linear increase in $K_\mathrm{f}$ going from $\ce{La^3+}$ to $\ce{Lu^3+}$ is entirely to be expected. The ionic radius decreases going across the 4f block, and consequently, the interactions with the ligand (primarily electrostatic in nature) become stronger.
However, as the title suggests, I'm interested in the slight blip at gadolinium ($\ce{Gd^3+}$). The value of $K_\mathrm{f}$ seems to be lower than would be expected, and the astute reader will notice that this applies not just to the EDTA complexes but also for some of the other ligands investigated.
Is there a reason for this anomaly?
There is a review by Moeller et al. from 1965,2 which wrote:
For all ligands which have been studied, the gadolinium complex is less stable than would be expected from the simple electrostatic model. This behavior has been called the “gadolinium break” and cannot be explained, as was originally attempted, by assuming a steric effect, since it is still apparent in ligands for which there should be no steric interference.
Of course, this is more than half a century ago. I would be grateful if anybody could provide either a sound theoretical explanation for why the Gd formation constants should be smaller or a newer reference that explains this effect.
References:
Smith, R. M.; Martell, A.E. Critical stability constants, enthalpies and entropies for the formation of metal complexes of aminopolycarboxylic acids and carboxylic acids. Sci. Total Environ. 1987, 64 (1–2), 125–147. DOI: 10.1016/0048-9697(87)90127-6.
Moeller, T.; Martin, D. F.; Thompson, L. C.; Ferrús, R.; Feistel, G. R.; Randall, W. J. The Coordination Chemistry of Yttrium and the Rare Earth Metal Ions. Chem. Rev. 1965, 65 (1), 1–50. DOI: 10.1021/cr60233a001.