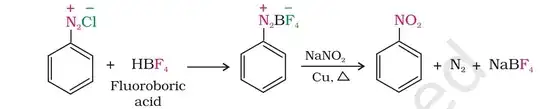
After a bit of research on benzenediazonium fluoroborate, I came up with an (complete?) answer although it is based on some approximations and/or speculations.
Benzenediazonium fluoborate is a versatile organic salt used to create various other organic compounds like by helping to introduce aryl, arylazo, arylhydrazono, or amino groups; forms fluoroarenes upon heating and are building block for heterocycles. They have a much higher stability than the corresponding chlorides and shock-insensitive. Generally decomposes at decomposes at 114-116 °C (Source).
So, what causes it much higher stability? Maybe it is their ion size. The benzenediazonium cation and fluoroborate anion are of same size resulting their stability[citation needed?]. Or maybe it is due to the resonance of benzenediazonium cation.
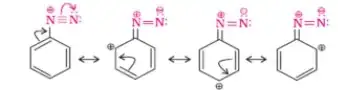
But when you apply heat to the salt, it is bound to decompose, maybe because of two reasons. One is that exceptionally stable nitrogen is formed on its decomposition (i.e nitrogen is formed from nitrogen cation) and second is the $\ce{N-F}$ bond is long and weak. From the information regarding structure of benzenediazonium fluorobotate, it is evident (Here):
Benzenediazonium tetrafluoroborate, $\ce{C6H5N2+•BF4−}$, crystallizes
in space group P21/a with unit cell dimensions a = 17.347(2),
b = 8.396(1), c = 5.685(1) Å, β = 92.14(1)°, Z = 4. The structure was
solved by direct phasing methods using the program SHELX 76. The
parameters were refined by full-matrix least-squares to a final
R = 0.063 for 1346 observed reflections. $\ce{C—N}$ and $\ce{N≡N}$
bond lengths are 1.415(3) and 1.083(3) Å, respectively, and the bonds
of the benzene ring vary from 1.371(5) to 1.383(4) Å. There are
three $\ce{N-F}$ close contacts of ≤ 2.84 Å and the positive charge
appears to be shared between the nitrogen atoms.
Below is the abstract of the mechanism of benzenediazonium tetrafluoroborate decomposition to give an insight (full pdf behind paywall is here):
The thermolysis of benzenediazonium tetrafluoroborate was studied by
thermogravimetry in dynamic mode. The decomposition of
$\ce{[ArNN]+BF4−}$ in the solid state with the formation of
$\ce{C6H5F, BF3, C6H6}$, and $\ce{N2}$ starts at T>348 K. The speed of
the thermolysis was estimated gravimetrically and by infrared
spectroscopy, considering the change of the intensity of the
absorption band at 1498 cm−1, which corresponds to fluorobenzene. The
maximal rate of thermolysis observes at the 366.5 K. A kinetic scheme,
which includes the formation of a neutral complex
$\ce{[C6H5δ+⋯BF4δ−]}$, is proposed for the thermolysis of
arenediazonium tetrafluoroborate. The decomposition of the complex
with the formation of free-radical intermediates explains the chain
character of the thermolysis.
There is large hydrophobic part in diazonium ion and in $\ce{BF4-}$ ion, size is large enough that hardly any hydration energy would be released to cause the dissolution. Hence the salt is insoluble. (Here)