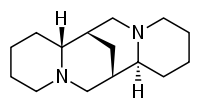
In its pure form (–)-sparteine is an only-slightly-yellow clear, viscous liquid. Yet, after only a week of being kept in dry, normal atmosphere (in a round bottom flask covered by a septa), some of it reacts with something to form a sticky, orange-brown substance. I've had to distill the sparteine I have twice due to whatever reaction is occurring, and I would like to know exactly what the reaction is so I don't have to distill every time I need it. The literature I found mentioned that it readily absorbs carbon dioxide in a 1-1 molar ratio, and I know that tertiary amines do sometimes react with CO2, but I haven't been able to piece it together as something that could happen in ambient conditions.