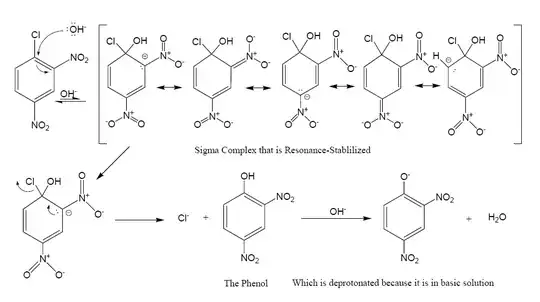
Yes, it's anionic sigma complex, more precisely Meisenheimer complex. They are much more stable then arenium ions (Wheland intermediates) and can be isolated much more easily.
There was actually synthesized a sigma complex which is a zwitterionic combination of both Meisenheimer and Wheland complexes, which shows how well they can be stabilised:
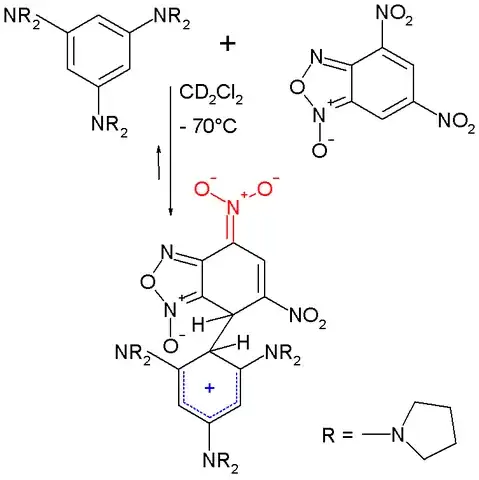
(picture created by Wikipedia User:V8rik)
The term "sigma complex" has also other meanings. Organometallic compounds with sigma bonds connecting with aromatic rings are called like that, too. There are also sigma complexes of cyclobutadiene with aluminium chloride.
Generally, molecules are called sigma complexes to stress the difference between them and pi complexes which can also be created from cyclic annulenes. First ones are bonded using sigma bonds, the other ones use pre-existing pi orbitals.