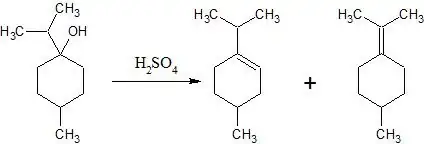
What will be the major product of this elimination reaction?
In the acid-catalyzed dehydration of 1-methylcyclohexanol, both 1-methylcyclohexene and methylenecyclohexane are formed in ca. an 85:15 ratio. In your example we have two more substituents on the exocyclic double bond which will serve to further stabilize the exocyclic double bond and increase the amount of the exocyclic isomer. I don't know for sure how much it will push the equilibrium towards the exocyclic isomer, but I might guess that the product mixture would be at least 50:50, if not more of the exocyclic isomer - but I would expect a mixture.
what influence will kinetics have on this reaction?
Well you won't see the kinetics unless you go out of your way and look for it. Factors that influence which isomer is kinetically favored include:
- statistical factors - there are 4 ring hydrogens that can be eliminated vs. only 1 on the isopropyl group; this favors kinetic formation of the endocyclic isomer
- alignment of the hydrogen with the p-orbital on the carbocation carbon - the hydrogen on the isopropyl group can better align with the p-orbital than those hydrogens in the ring, however the cyclohexane ring can easily distort; so while this isn't a major factor it favors the exocyclic isomer
- steric factors - I agree with your analysis, but would guess that the difference is small; still sterics would slightly favor the endocyclic isomer
It seems pretty close to call, but if I had to guess I'd say that the statistical factor wins out (and the steric argument further helps) and the endocyclic isomer would likely be the the kinetic product.
But again, in acid this is a fast equilibrium; the products will quickly equilibrate and the thermodynamically controlled product distribution will be observed. Maybe if you run the reaction at low temperature with a strong dehydrating agent ($\ce{POCl3}$) you could capture some meaningful kinetic information.