In the electrophilic aromatic substitution (EAS) of pyrrole, why is the alpha position preferred over the beta position so much?
Asked
Active
Viewed 1.3k times
1 Answers
18
The following figure shows the resonance structures we can draw to describe the intermediates produced by electrophilic at the 2- ($\alpha$) or 3- ($\beta$) positions in pyrrole.
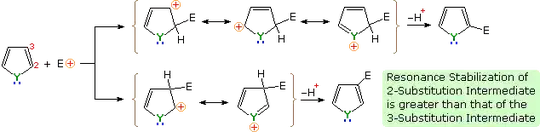
You can see that the intermediate produced by attack at the $\alpha$-position can be described by 3 resonance structures. Whereas attack at the $\beta$-position produces an intermediate that can be described by only 2 resonance structures.
Therefore, the intermediate formed by $\alpha$ attack is more stable and the activation energy leading to it will be reduced. Consequently, electrophilic attack at the $\alpha$ position in pyrrole is preferred.
orthocresol
- 71,033
- 11
- 239
- 410
ron
- 84,691
- 13
- 231
- 320