It is common that adding common salt into water will lower its melting point. But, is there any way to RAISE the melting point of water? Does it lower the boiling point of water as well? Why?
4 Answers

One way to raise the melting point of water is to increase pressure beyond about 635 MPa. By raising pressure you could get the melting point to be even greater than the normal boiling point.
A second way is to lower the pressure, but this can only increase the melting point by 0.01 K.
A third way would be to added enough of a high melting point solute that you pass the eutectic point. In other words, generally when you add a solute to water, the melting point decreases, but only until you add enough solute that the eutectic point is reached, beyond which adding more solute increases the melting point.
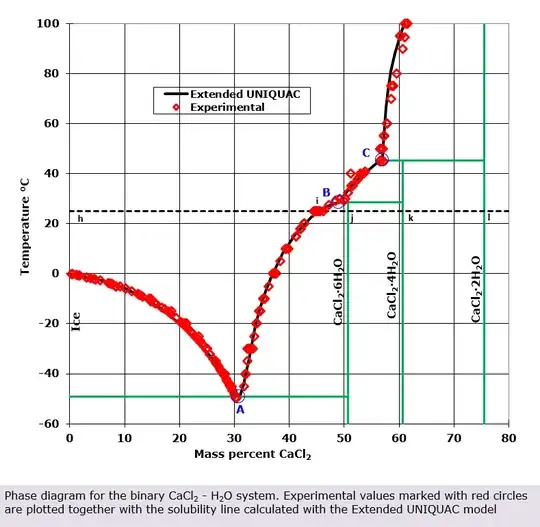
source: http://www.phasediagram.dk/binary/CaCl2.PNG
For example, in the diagram, above, A is the eutectic point. Beyond this point, adding more CaCl2 increases freezing point. Then beyond 38% CaCl2, melting point exceeds ) degrees C.
- 40,570
- 2
- 85
- 181
You can do it with methane under pressure (but more moderate pressure than in @DavePhD's answer) (https://en.wikipedia.org/wiki/Methane_clathrate). Methane is more soluble in the caged clathrate structure than in liquid water, so the former is stabilized over the latter. For example, at 50 atm (5 MPa) methane pressure the melting point is raised to $+4°C$.
Such clathrates complicate natural gas processing, an issue also identified in the reference cited above.
- 56,895
- 4
- 89
- 175
Just remember 'water' will not freeze at hypereutectic concentration above liquidus point. Below solidus point, you still won't get ice, only precipitate of the salt.
- 1
-
If what you want is for the water to solidify into ice at a higher temperature, it is impossible to do by adding solute to water, or indeed any other substances. – Eran Feb 22 '17 at 23:41
-
Nope. The water still freezes as part of the eutectic mixture after the excess solute has precipitated out. This eutectic freezing occurs at the eutectic point and does not qualify as raising the melting point of water. You can raise the melting point with a few solutes that are more soluble in the solid phase than the liquid; methane under pressure does that as described in another answer. – Oscar Lanzi May 01 '18 at 16:35
Water is a liquid, thus already melted. Your question should either be about raising the melting point of ice or raising the boiling point of water.
To raise the melting point of ice, meaning trying to keep ice as a solid at a temperature as high as possible, reduce impurities.
To raise the boiling point of water, meaning trying to keep water liquid at a temperature as high as possible, increase the pressure, that is, do it in a pressure cooker instead of with a surface exposed to atmospheric pressure.
- 115
- 2
- 2
- 8
-
5Your first statement is obnoxiously pedantic and wrong. Yes there is a special word for frozen water, but it's still water. It goes without saying that we are talking about melting ice if we say 'melting water'. – ringo Feb 23 '17 at 05:27
-
2
From: Wikipedia
why is there liquid solution beyond 23% salt solution and below 0 degree Celsius?
– Chin Huan Feb 16 '15 at 09:20