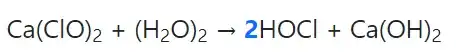Let imagine there are 3 bank accounts, one with 10000 Euros, one with 10000 US dollars, one with 10000 UK pounds. Let assume - just illustratively - there is exchange ratio 5 dollars per 4 Euros and 2 dollars per 1 UK pound. If there is requirement to express the money level "as dollars",
the first one is worthy "as 12500 dollars",
the third one "as 20000 dollars".
Back to chemistry, "as Cl2" is not limited to chlorine being a part of the considered compound. there is just expected some chemical equivalence, defined by chemical reactions.
$\pu{1 mol}$ $\ce{Cl2}$ is equivalent to $\pu{1 mol}$ of $\ce{HClO}$ or $\ce{ClO-}$ due the reactions
$$\ce{Cl2(aq) + H2O <=> H+(aq) + Cl-(aq) + HClO(aq)}$$
$$\ce{Cl2(aq) + 2 OH-(aq) <=>> ClO-(aq) + Cl-(aq) + H2O}$$
So if
$M_\ce{Cl2} \approx \pu{71 g mol-1}$
$M_\ce{HClO} \approx \pu{52.5 g mol-1}$
then $\pu{52.5 g}$ $\ce{HClO}$ means "as $\ce{Cl2}$" $\pu{71 g} \ce{Cl2}$.
Mass-wise, it does not matter if we consider $\ce{Cl}$ or $\ce{Cl2}$). Amount-wise, $\ce{HClO}$ is equivalent to twice as much amount of $\ce{Cl}$.
If we consider reactions
$$\ce{2 Fe^2+(aq) + H2O2(aq) + 2 H+(aq) -> 2 Fe^3+(aq) + 2 H2O}$$
$$\ce{2 Fe^2+(aq) + Cl2(aq) -> 2 Fe^3+(aq) + 2 Cl-(aq)}$$
then $\pu{1 mol}$ of $\ce{Cl2}$ is equivalent $\ce{1 mol}$ of $\ce{H2O2}$, so $\pu{34 g}$ of $\ce{H2O2}$, if expressed "as $\ce{Cl2}$", means (approximately) $\pu{71 g}$ $\ce{Cl2}$.
The notation mentioned in the other thread ($\ce{NH3-N}$) means something little different: "nitrogen in ammoniacal form, expressed as elemental nitrogen.". The direct equivalent, if we e.g. analyzed solution containing chloride $\ce{Cl-}$, hypochlorite $\ce{ClO-}$ and chlorate $\ce{ClO3-}$, would be the analogical syntax $\ce{Cl-Cl}$, $\ce{ClO-Cl}$, $\ce{ClO3-Cl}$, or verbose equivalents "chlorine in chloride/hypochlorite/chlorate form.".
It is not used to express equivalence of different chemicals, but rather to express content of chemical element (or compound), present in various forms, in a unified way. Typically to make their sum easily.
Like in elemental analysis, the mass of total nitrogen in a waste water sample is the sum of ammoniacal+nitrite+nitrate+organic nitrogen.
$$m_\text{Total-N}=m_\ce{NH3-N}+m_\ce{NO2-N}+m_\ce{NO3-N}+m_\ce{Org-N}$$
If we consider various chloramins:
\begin{align}
\ce{1 NH2Cl} \equiv \ce{1 HOCl} \equiv \ce{1 Cl2}\\
\ce{1 NHCl2} \equiv \ce{2 HOCl} \equiv \ce{2 Cl2}\\
\ce{1 NCl3} \equiv \ce{3 HOCl} \equiv \ce{3 Cl2}
\end{align}
As
\begin{align}
\ce{NH_{3-n}Cl_n + n H2O &<=> NH3 + n HOCl}\\
\ce{HOCl + Cl- + H+ &<=> H2O + Cl2}
\end{align}
If we consider $\ce{NHCl2-Cl}$ as "chlorine in form of dichloramin,
$$\pu{1 mg L-1} \ce{NHCl2} \equiv \frac{2 \cdot M_\ce{Cl}}{M_\ce{NHCl2}} \pu{mg L-1} \ce{NHCl2-Cl}$$
But, if we consider $\ce{NHCl2}$ "as $\ce{Cl2}$, or as active chlorine",
$$\pu{1 mg L-1} \ce{NHCl2} \equiv \frac{4 \cdot M_\ce{Cl}}{M_\ce{NHCl2}} \pu{mg L-1} \ce{Cl}$$
- The former counts the equivalent mass concentration for the chlorine fraction contained in the substance.
- The latter counts the mass concentration for the equivalent free/active chlorine.