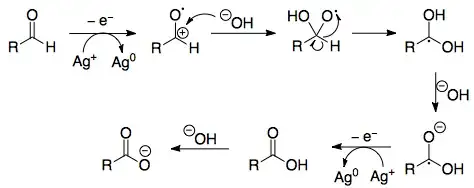
The balanced equation can tell you a lot. This is the balanced equation:
$\ce{R-CHO + 2[Ag(NH3)2]+ + 3HO- -> R-COO- + 2Ag + 2H2O + 4NH3}$
Let's check out what's happening:
1) First, the aldehyde is being oxidized. Specifically, the carbon in the aldehyde is being oxidized; it is losing electrons to silver.
Oxidation half reaction: $\ce{R-CHO -> R-COO- + 2e^- + H+}$
Oxidation states of C: $~~~~~~+1~~~~~~~~~~~~~+3$
2) Mass isn't balanced; we need a source of $\ce{O^2-}$ (negative 2 oxidation state oxygen atoms). Given that this reaction is happening with a solution of silver ammonia, it makes thermodynamic sense that your best source of $\ce{O^2-}$ oxygen is the hydroxide anion: $\ce{HO^-}$.
Water is a possible source but it's pricier to heterolytically cleave two $\ce{H-O}$ bonds as opposed to just one $\ce{H-O}$ bond.
Thermodynamically favorable: $\ce{H-O^- -> H^+ +O^2-}$
Not so thermodynamically favorable: $\ce{H-O-H -> 2H^+ +O^2-}$
3) The freeing of one $\ce{O^2-}$ yields the hydrogen proton, $\ce{H+}$. This is an acid; it will react immediately with the strongest base in the system, which would be the hydroxide anion.
Also, note that a hydrogen proton is freed from the aldehyde. The hydrogen atom is freed heterolytically as to yield 2 electrons, which go to the silver ammonia complex.
$\ce{R-CHO -> R-COO- + H+ + 2e^-}$
Two protons require two additional hydroxide anions to react with; this is why we use a total of three hydroxide anions and this yields two water molecules.
$\ce{2H+ + 2HO- -> 2H2O}$
4) The above explains the hard stuff. The reduction of silver is easy:
$\ce{[Ag(NH3)_2]+ + e^- -> Ag + 2NH3}$
The silver in the silver ammonia complex ion has an oxidation state of +1; gaining an electron allows solid silver to precipitate out, giving a positive Tollens test for aldehydes. The ammonia remains unchanged.