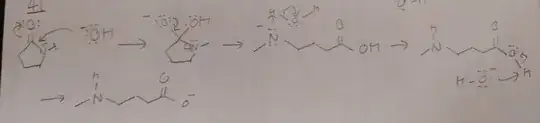In this mechanism, I got a pretty different mechanism from the solution. From the model answer, there is an intramolecular proton transfer just before the formation of the carboxylate ion. However, in the second intermediate, I think the electrostatic repulsion between nitrogen and oxygen is large, increase separation between nitrogen and hydrogen, hence reducing chance of intramolecular transfer.
My purposed mechanism rely on the H2O given in reaction mixture to undergo proton transfer, forming OH- (matching basic condition) and then pull the proton from R-COOH to form the carboxylate salt (product).
Is my mechanism plausible? What mistakes I have made if the model answer is the only correct mechanism?
Also, please suggest ways that I can improve this type of questions, thank you very much.