Hi I am trying to figure out the mechanism of a reaction containing a lithium carbanion and 1,2-dibromoethane in THF. The lithium carbanion is generated by deprotonating using LDA. Then 1,2-dibromoethane is added to get the desired product (see image)
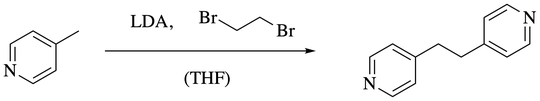
I think that the carbanion is brominated, otherwise this reaction does not make sense, but I really don't understand the mechanism. 1,2-dibromoethane can undergo a reductive elimination of the bromide-ions to form bromine and ethene. But in this specific reaction, I have no reductive agent... So how does the carbanion get brominated? Is it simply a fragmentation reaction, where the carbanion attacks the Br of 1,2-dibromoethane?

