When a heavy nucleus undergoes nuclear fission , it splits to two smaller, more stable nuclei and produces heat, similarly, when two light nuclei fuse, to make a heavier, more stable nucleus and produces energy.
That is correct. But note that there is a distinction between "light nuclei" and "heavy nuclei."
In other words "light nuclei" are light because when they fuse they release energy. But this doesn't say that you can keep fusing the "light products" ad infinitum and that you will keep getting another "light" nucleus. (Think about two mass 4 atoms fusing to make a mass 8 nucleus. Then two mass 8 atoms fusing to make a mass 16 nucleus, and so on....)
And "heavy nuclei" are heavy because when they fuse they release energy. But this doesn't say that you can keep splitting the "less heavy products" ad infinitum and that you will keep getting another "heavy" nucleus. (Think about a mass 256 atoms splitting to make two mass 128 nucleus. Then a mass 128 atoms splitting to make two mass 64 nucleus, and so on....)
The nuclear binding energy curve is shown below.
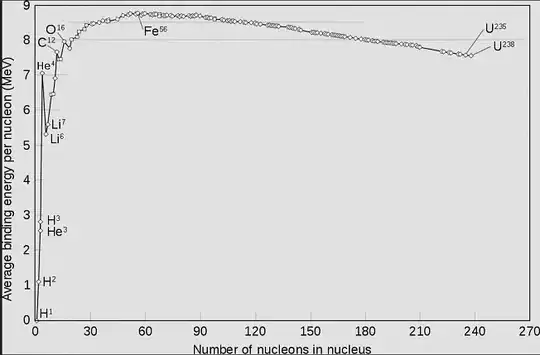
So in a star fusing light elements to form heavier isotopes releases energy until $\ce{^{56}Fe}$. To make heavier elements requires energy. Thus elements heavier than $\ce{^{56}Fe}$ are not made by stellar fusion, but from super nova explosions and the collision of neutron starts.
